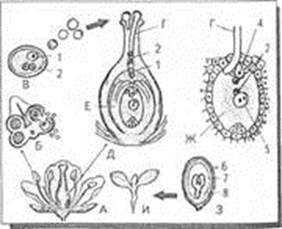
8. Read text B and choose the best title for it:
Zone-refining.
Impurity atoms.
3. The elimination of crystal defects.
Make sure that you know the following words and word combinations:
Extreme purification - экстремальная очистка
corrosion resistance - устойчивость к коррозии
refining – рафинирование
the coil – катушка
impurity atoms - атомы примеси
Text B
Extreme purification of a metal can radically improve properties such as ductility, strength and corrosion resistance. Zone-refining was devised by W. G. Pfann, its development being ‘driven’ by the demands of the newly invented transistor for homogeneous and ultrapure metals (e.g. Si, Ge). The method takes advantage of non-equilibrium effects associated with the ‘pasty’ zone separating the liquidus and solidus of impure metal. Considering the portion of Figure 3.12 where addition of solute lowers the liquidus temperature, the concentration of solute in the liquid,  , will always be greater than its concentration
, will always be greater than its concentration  in the solid phase; that is, the distribution coefficient k =
in the solid phase; that is, the distribution coefficient k =  /
/  is less than unity. If a bar of impure metal is threaded through a heating coil and the coil is slowly moved, a narrow zone of melt can be made to progress along the bar. The first solid to freeze is purer than the average composition by a factor of k, while that which freezes last, at the trailing interface, is correspondingly enriched in solute. A net movement of impurity atoms to one end of the bar takes place. Repeated traversing of the bar with a set of coils can reduce the impurity content well below the limit of detection (e.g. <1 part in
is less than unity. If a bar of impure metal is threaded through a heating coil and the coil is slowly moved, a narrow zone of melt can be made to progress along the bar. The first solid to freeze is purer than the average composition by a factor of k, while that which freezes last, at the trailing interface, is correspondingly enriched in solute. A net movement of impurity atoms to one end of the bar takes place. Repeated traversing of the bar with a set of coils can reduce the impurity content well below the limit of detection (e.g. <1 part in  for germanium). Crystal defects are also eliminated: Pfann reduced the dislocation density in metallic and semi-metallic crystals from about 3.5
for germanium). Crystal defects are also eliminated: Pfann reduced the dislocation density in metallic and semi-metallic crystals from about 3.5 

 to almost zero. Zone-refining has been used to optimize the ductility of copper, making it possible to cold-draw the fine-gauge threads needed for interconnects in very large-scale integrated circuits.
to almost zero. Zone-refining has been used to optimize the ductility of copper, making it possible to cold-draw the fine-gauge threads needed for interconnects in very large-scale integrated circuits.
Part V
1. Learn the words:
| intermediate
| промежуточный
|
| stoichiometric ratios
| стехиометрические соотношения
|
| constituent atoms
| составляющие атомы
|
| advisable
| рекомендуемый
|
| to avoid
| избегать
|
| to extend
| простираться
|
| ordinate
| ордината
|
| hard and brittle
| жесткий и хрупкий
|
| to restrict
| ограничивать
|
| to solder
| паять, припаять
|
| data
| данные
|
| distribution
| распределение
|
| lamella
| ламель
|
| spheroid
| сфероид
|
| intergranular films
| межзерновые пленки
|
| feature
| особенность
|
| the surface energy
| поверхностная энергия
|
| the strain energy
| энергия деформации
|
| to disperse
| рассеивать
|
| evenly
| равномерно
|
| boundary surface
| граничная поверхность
|
| sulphide particles
| сульфидные частицы
|
| manganese sulphide
| сульфид марганца
|
| tolerable
| терпимый, допустимый
|
| sulphide films
| сульфидные пленки
|
| embrittlement
| охрупчивание
|
| quenching
| гашение, закалка
|
| martensite and bainite
| мартенсит и бейнит
|
| to devise
| изобретать, разрабатывать
|
| pearlite
| перлит
|
| the orthorhombic intermediate phase
| орторомбическая промежуточная фаза
|
| a lamellar composite of soft, ductile ferrite
| пластинчатый композит из мягкого, пластичного феррита
|
| to predate
| предшествовать
|
| carbon
| углерод
|
| to dissolve
| растворяться
|
| interstitial
| интерстициальный, промежуточный
|
| versatile behaviour of steels
| универсальное поведение сталей
|
2. Guess the meaning of the following words and word combinations:
A phase diagram; compound; room temperature; melt or freeze; to be congruent (incongruent); a congruently melting phase; binary or ternary; compositional data; the most serious limitation; to distribute, distributor, distribution; volume and shape; behavior of materials; properties of the material; equilibrium states; temperature-dependency; allotropes.
3. Identify the part of speech and translate the words:
To differ, different, difference; to appear, appearance; frequent, frequently, frequency; advisable, advisability, advisably; to extend, extended, extending; extreme, extremely; to interpret, interpreter, interpretation; to depend, dependent, dependable; to disperse, dispersed.
4. Match the words with their definitions:
| a single vertical line in the diagram
| разделить сложную фазовую диаграмму
|
| melt or freeze without any change in composition
| понятные детали
|
| a melting point
| простые эвтектические подсистемы
|
| a convenient means
| медные сплавы
|
| to divide a complex phase diagram
| нежелательные хрупкие слои
|
| understandable parts
| удобное средство
|
| simple eutectic sub-systems
| точка плавления
|
| copper alloys
| расплавляться или замораживаться без изменения состава
|
| undesirable brittle layers
| одна вертикальная линия на диаграмме
|
Text A
Intermediate phases
An intermediate phase differs in crystal structure from the primary phases and lies between them in a phase diagram. In Figure 3.19, which shows the diagram for the Mg–Si system,  Si is the intermediate phase. Sometimes intermediate phases have definite stoichiometric ratios of constituent atoms and appear as a single vertical line in the diagram. However, they frequently exist over a range of composition and it is therefore generally advisable to avoid the term ‘compound’. In some diagrams, such as Figure 3.19, they extend from room temperature to the liquidus and melt or freeze without any change in composition. Such a melting point is said to be congruent: the melting point of a eutectic alloy is incongruent. A congruently melting phase provides a convenient means to divide a complex phase diagram (binary or ternary) into more readily understandable parts. For instance, an ordinate through the vertex of the intermediate phase in Figure 3.19 produces two simple eutectic sub-systems. Similarly, an ordinate can be erected to pass through the minimum (or maximum) of the liquidus of a solid solution (Figure 3.38b). In general, intermediate phases are hard and brittle, having a complex crystal structure (e.g.
Si is the intermediate phase. Sometimes intermediate phases have definite stoichiometric ratios of constituent atoms and appear as a single vertical line in the diagram. However, they frequently exist over a range of composition and it is therefore generally advisable to avoid the term ‘compound’. In some diagrams, such as Figure 3.19, they extend from room temperature to the liquidus and melt or freeze without any change in composition. Such a melting point is said to be congruent: the melting point of a eutectic alloy is incongruent. A congruently melting phase provides a convenient means to divide a complex phase diagram (binary or ternary) into more readily understandable parts. For instance, an ordinate through the vertex of the intermediate phase in Figure 3.19 produces two simple eutectic sub-systems. Similarly, an ordinate can be erected to pass through the minimum (or maximum) of the liquidus of a solid solution (Figure 3.38b). In general, intermediate phases are hard and brittle, having a complex crystal structure (e.g.  C, Cu
C, Cu  (ϴ)). For instance, it is advisable to restrict time and temperature when soldering copper alloys, otherwise it is possible for undesirable brittle layers of
(ϴ)). For instance, it is advisable to restrict time and temperature when soldering copper alloys, otherwise it is possible for undesirable brittle layers of  Sn and
Sn and  to form at the interface.
to form at the interface.
Limitations of phase diagrams. Phase diagrams are extremely useful in the interpretation of metallic and ceramic structures but they are subject to several restriction. Primarily, they identify which phases are likely to be present and provide compositional data. The most serious limitation is that they give no information on the structural form and distribution of phases (e.g. lamellae, spheroids, intergranular films, etc.). This is unfortunate, since these two features, which depend upon the surface energy effects between different phases and strain energy effects due to volume and shape changes during transformations, play an important role in the mechanical behavior of materials. This is understood if we consider a two phase (α+β) material containing only a small amount of β-phase. The β-phase may be dispersed evenly as particles throughout the α-grains, in which case the mechanical properties of the material would be largely governed by those of the α-phase. However, if the β - phase is concentrated at grain boundary surfaces of the α-phase, then the mechanical behaviour of the material will be largely dictated by the properties of the β-phase. For instance, small amounts of sulphide particles, such as grey manganese sulphide (MnS), are usually tolerable in steels but sulphide films at the grain boundaries cause unacceptable embrittlement. A second limitation is that phase diagrams portray only equilibrium states. As indicated in previous sections, alloys are rarely cooled or heated at very slow rates. For instance, quenching, as practised in the heat-treatment of steels, can produce metastable phases known as martensite and bainite that will then remain unchanged at room temperature. Neither appears in phase diagrams. In such cases it is necessary to devise methods for expressing the rate at which equilibrium is approached and its temperature-dependency.
5. Open the brackets and put the predicate in the correct form, translate the sentences:

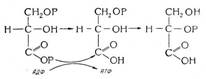



 , will always be greater than its concentration
, will always be greater than its concentration  in the solid phase; that is, the distribution coefficient k =
in the solid phase; that is, the distribution coefficient k =  for germanium). Crystal defects are also eliminated: Pfann reduced the dislocation density in metallic and semi-metallic crystals from about 3.5
for germanium). Crystal defects are also eliminated: Pfann reduced the dislocation density in metallic and semi-metallic crystals from about 3.5 

 to almost zero. Zone-refining has been used to optimize the ductility of copper, making it possible to cold-draw the fine-gauge threads needed for interconnects in very large-scale integrated circuits.
to almost zero. Zone-refining has been used to optimize the ductility of copper, making it possible to cold-draw the fine-gauge threads needed for interconnects in very large-scale integrated circuits. Si is the intermediate phase. Sometimes intermediate phases have definite stoichiometric ratios of constituent atoms and appear as a single vertical line in the diagram. However, they frequently exist over a range of composition and it is therefore generally advisable to avoid the term ‘compound’. In some diagrams, such as Figure 3.19, they extend from room temperature to the liquidus and melt or freeze without any change in composition. Such a melting point is said to be congruent: the melting point of a eutectic alloy is incongruent. A congruently melting phase provides a convenient means to divide a complex phase diagram (binary or ternary) into more readily understandable parts. For instance, an ordinate through the vertex of the intermediate phase in Figure 3.19 produces two simple eutectic sub-systems. Similarly, an ordinate can be erected to pass through the minimum (or maximum) of the liquidus of a solid solution (Figure 3.38b). In general, intermediate phases are hard and brittle, having a complex crystal structure (e.g.
Si is the intermediate phase. Sometimes intermediate phases have definite stoichiometric ratios of constituent atoms and appear as a single vertical line in the diagram. However, they frequently exist over a range of composition and it is therefore generally advisable to avoid the term ‘compound’. In some diagrams, such as Figure 3.19, they extend from room temperature to the liquidus and melt or freeze without any change in composition. Such a melting point is said to be congruent: the melting point of a eutectic alloy is incongruent. A congruently melting phase provides a convenient means to divide a complex phase diagram (binary or ternary) into more readily understandable parts. For instance, an ordinate through the vertex of the intermediate phase in Figure 3.19 produces two simple eutectic sub-systems. Similarly, an ordinate can be erected to pass through the minimum (or maximum) of the liquidus of a solid solution (Figure 3.38b). In general, intermediate phases are hard and brittle, having a complex crystal structure (e.g.  C, Cu
C, Cu  (ϴ)). For instance, it is advisable to restrict time and temperature when soldering copper alloys, otherwise it is possible for undesirable brittle layers of
(ϴ)). For instance, it is advisable to restrict time and temperature when soldering copper alloys, otherwise it is possible for undesirable brittle layers of  Sn and
Sn and  to form at the interface.
to form at the interface.