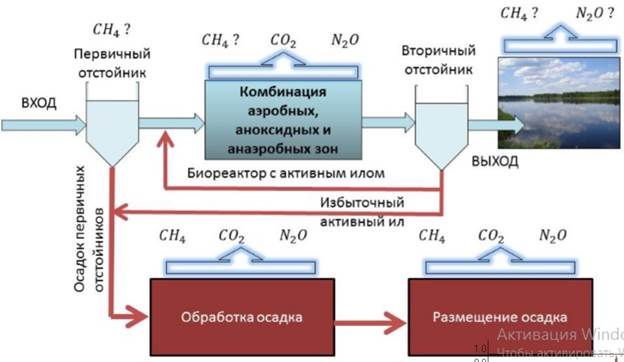
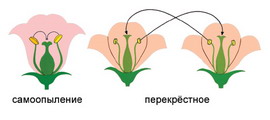
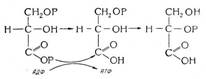
By Mark S. Lesney
Elsevier Global Medical News
Because anticoagulation is a balancing act between the prevention of clots and the risk of major bleeds, tremendous efforts have been made to find the safest, most effective drugs possible. The foundations of anticoagulant therapy – warfarin (taken orally) and heparin (taken intravenously) – are being shaken by the onslaught of the newer oral factor Xa inhibitors and a direct thrombin inhibitor.
Two of these inhibitors are currently approved by the Food and Drug Administration, with others poised for approval.
Warfarin, the current standard for outpatient oral anticoagulant therapy, leaves a lot to be desired. The need for International Normalized Ratio (INR) monitoring has made patient compliance a significant problem, as some patients must travel routinely to their doctor’s office or warfarin clinics for blood monitoring (although there has been some movement toward putting the INR-monitoring process into patient hands). Even for compliant patients, reaching and maintaining an appropriate INR can be difficult, and warfarin carries the risk of a major bleed, especially among fragile elderly patients.
But is a move from warfarin to the new drugs premature? Many concerns remain, including potential difficulties with surgery in patients taking these agents.
 The two FDA-approved drugs are rivaroxaban (Xarelto, marketed by Janssen Pharmaceuticals), which is a direct factor Xa inhibitor, and dabigatran (Pradaxa, marketed by Boehringer Ingelheim), a direct thrombin inhibitor.
The two FDA-approved drugs are rivaroxaban (Xarelto, marketed by Janssen Pharmaceuticals), which is a direct factor Xa inhibitor, and dabigatran (Pradaxa, marketed by Boehringer Ingelheim), a direct thrombin inhibitor.
Rivaroxaban (a member of the “xaban” family) was approved for two separate indications: the prevention of deep vein thrombosis in knee or hip replacement surgery (July 2011) and stroke prophylaxis in patients with nonvalvular atrial fibrillation (November 2011). Taken as a once-daily dose, the drug does not require routine blood monitoring – a key advantage over warfarin. According to the ROCKET-AF trial on which the approval was based, rivaroxaban had a major bleeding rate similar to that of warfarin; however, while it caused less bleeding into the brain, it caused more bleeding into the stomach and intestines.
During the approval process for rivaroxaban, FDA panelists questioned whether the drug was properly compared with the highest contemporary standards for warfarin and noted that it did not provide a “robust noninferiority” to dabigatran. These concerns led the panel to suggest that rivaroxaban be used only as a third-line therapy when warfarin and dabigatran are not options. Panelists also wondered what happens when patients stop using the drug. Rivaroxaban is now marketed with a boxed warning that sudden discontinuation increases the risk of stroke.
The FDA approved dabigatran in October 2010 for long-term anticoagulation in patients with atrial fibrillation, and by late August 2011, U.S. physicians had written 250,000 prescriptions for the drug.
But questions about the drug’s safety have been raised recently. In December 2011, the FDA announced that it was looking into reports of serious bleeding events associated with dabigatran, while reaffirming its belief that “the benefit of Pradaxa continues to exceed the potential risks when the drug is used appropriately.” The FDA’s concerns were prompted in part by a European Medicines Agency (EMA) safety update in November noting “a worldwide total of 256 spontaneous case reports of serious bleeding resulting in death.”
In October, the EMA’s Committee for Medicinal Products for Human Use had recommended that dabigatran be labeled to advise testing for renal function in all patients before starting treatment and, for those taking the drug, renal function assessment at least once a year in patients older than 75 years and in patients of any age with a suspected decline in renal function.
A meta-analysis of seven randomized clinical trials published online in the Archives of Internal Medicine (Arch. Intern. Med. 2012 Jan. 9 [doi:10.1001/archinternmed.2011.1666]) showed that dabigatran appeared to increase the risk of myocardial infarction and acute coronary syndrome, compared with warfarin, enoxaparin, and placebo. The incidence of these events was 1.19% in those taking dabigatran vs. 0.79% in the controls.
 Another new anticoagulant, apixaban, has been approved as Eliquis in the European Union and fast-tracked for approval in the United States. Apixaban outperformed warfarin in the ARISTOTLE trial in preventing stroke, reducing bleeding, and increasing survival, regardless of how well controlled patients were on warfarin, dabigatran, and rivaroxaban. Results achieved with apixaban were better than those seen in the trials of dabigatran and rivaroxaban against warfarin (N. Engl. J. Med 2011;365:981-92).
Another new anticoagulant, apixaban, has been approved as Eliquis in the European Union and fast-tracked for approval in the United States. Apixaban outperformed warfarin in the ARISTOTLE trial in preventing stroke, reducing bleeding, and increasing survival, regardless of how well controlled patients were on warfarin, dabigatran, and rivaroxaban. Results achieved with apixaban were better than those seen in the trials of dabigatran and rivaroxaban against warfarin (N. Engl. J. Med 2011;365:981-92).
Of particular interest is apixaban’s good bleeding profile, compared with those of the other drugs (N. Engl. J. Med. 2011;364:806-17). “We have two trials in a large program showing this safety. In AVERROES, the bleeding risk with apixaban was the same as with low-dose aspirin,” Dr. Lars Wallentin said in an interview. Dr. Wallentin is professor and head of cardiology research at Uppsala (Sweden) University and a coinvestigator in the ARISTOTLE trial.
The APPRAISE-2 trial showed that adding apixaban to standard antiplatelet therapy in patients with acute cardiac syndrome resulted in a 1.3% rate of major bleeding, compared with 0.5% with placebo, including five fatal bleeding events vs. none with placebo.
Unlike warfarin, these new anticoagulants have no defined antidotes – a lack that is of particular concern to surgeons. In a review of the new antithrombotic drugs presented at the 2011 VEITH symposium, Dr. Russell H. Samson said of dabigatran: “There is no antidote, so this drug should be used sparingly if surgery is anticipated. Thus, it is probably not a drug that should be used to prevent graft failure.” Dr. Samson is a clinical associate professor of vascular surgery at Florida State University in Tallahassee.
A recent editorial in the New England Journal of Medicine sounded another note of caution. “Switching to a newer agent may not be necessary for the individual patient in whom INR has been well controlled with warfarin for years,” Dr. Jessica Mega of Brigham and Women’s Hospital, Boston wrote (N. Engl. J. Med. 2011;365:1052-4). “In addition, although the newer anticoagulants have a more rapid onset and termination of anticoagulation than does warfarin, agents to reverse the effects of the drugs are still under development and are not routinely available.”
The enthusiasm for the new anticoagulants may be premature for other reasons as well, such as the cost-effectiveness of these drugs, compared with the much cheaper warfarin, Dr. Mega said.
Direct thrombin inhibitors (bivalirudin, dabigatran, etc.) and direct Xa inhibitors (rivaroxaban) are being used with increasing frequency, so an “adequate” knowledge of anticoagulation must now include these newer agents, said Dr. Ravi Rajani, who was asked to comment on this report.
“Every midlevel surgical resident knows how to dose heparin and how to reverse warfarin, but many practicing surgeons have never heard of rivaroxaban or dabigatran until they are forced to operate on a patient who is taking them,” said Dr. Rajani, assistant professor of surgery at Emory University and director of vascular and endovascular surgery at Grady Memorial Hospital, both in Atlanta.

 “Furthermore, it is important to remember that these newer anticoagulants have been FDA approved for very specific indications only. We are starting to see off-label use of dabigatran as an end-all substitute for warfarin, without the evidence to really support it as such. Until we have more experience with these agents, their usage should be reserved for approved indications only,” he added.
“Furthermore, it is important to remember that these newer anticoagulants have been FDA approved for very specific indications only. We are starting to see off-label use of dabigatran as an end-all substitute for warfarin, without the evidence to really support it as such. Until we have more experience with these agents, their usage should be reserved for approved indications only,” he added.
Dr. Rajani said he had no relevant disclosures.
Elizabeth Mechcatie, Kerri Wachter, and Mitchel L. Zoler contributed to this report.
21.03
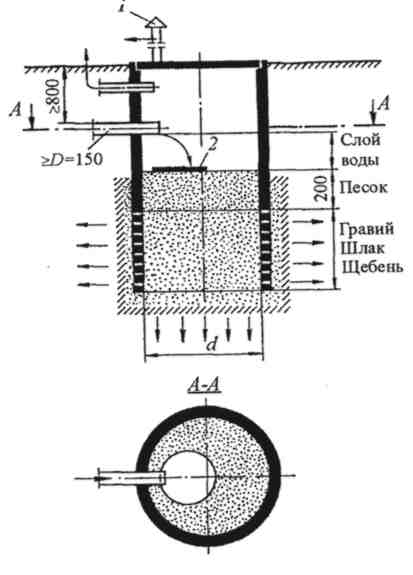
1. Ссылки на иностранные материалы по
«Протоколу Милуоки»
1) www.rabiesinfection.com/rabies-treatment/milwaukee-pr..
Сайт посвященный Бешенству, где дается анализ данного заболевания, его симптомов, методов лечения и профилактики, его распространения.
2) www.chw.org/display/PPF/DocID/33223/router.asp
Сайт Детской Больницы шт. Висконсин, США. На сайте представлены документы в формате PDF, содержащие материалы Протокола Милуоки, исследования, посвященные данному методу лечения бешенства, а так же краткие анкеты «ранняя симптоматика бешенства».

 The two FDA-approved drugs are rivaroxaban (Xarelto, marketed by Janssen Pharmaceuticals), which is a direct factor Xa inhibitor, and dabigatran (Pradaxa, marketed by Boehringer Ingelheim), a direct thrombin inhibitor.
The two FDA-approved drugs are rivaroxaban (Xarelto, marketed by Janssen Pharmaceuticals), which is a direct factor Xa inhibitor, and dabigatran (Pradaxa, marketed by Boehringer Ingelheim), a direct thrombin inhibitor.
 Another new anticoagulant, apixaban, has been approved as Eliquis in the European Union and fast-tracked for approval in the United States. Apixaban outperformed warfarin in the ARISTOTLE trial in preventing stroke, reducing bleeding, and increasing survival, regardless of how well controlled patients were on warfarin, dabigatran, and rivaroxaban. Results achieved with apixaban were better than those seen in the trials of dabigatran and rivaroxaban against warfarin (N. Engl. J. Med 2011;365:981-92).
Another new anticoagulant, apixaban, has been approved as Eliquis in the European Union and fast-tracked for approval in the United States. Apixaban outperformed warfarin in the ARISTOTLE trial in preventing stroke, reducing bleeding, and increasing survival, regardless of how well controlled patients were on warfarin, dabigatran, and rivaroxaban. Results achieved with apixaban were better than those seen in the trials of dabigatran and rivaroxaban against warfarin (N. Engl. J. Med 2011;365:981-92).
 “Furthermore, it is important to remember that these newer anticoagulants have been FDA approved for very specific indications only. We are starting to see off-label use of dabigatran as an end-all substitute for warfarin, without the evidence to really support it as such. Until we have more experience with these agents, their usage should be reserved for approved indications only,” he added.
“Furthermore, it is important to remember that these newer anticoagulants have been FDA approved for very specific indications only. We are starting to see off-label use of dabigatran as an end-all substitute for warfarin, without the evidence to really support it as such. Until we have more experience with these agents, their usage should be reserved for approved indications only,” he added.