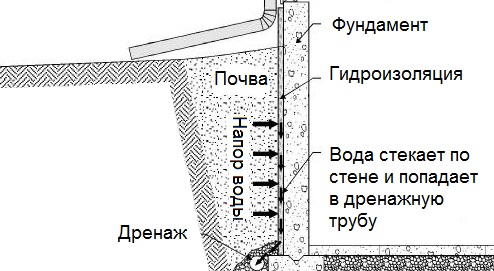
П.з.
Antoine-Laurent de Lavoisier (26 August 1743 – 8 May 1794) was a French nobleman and chemist central to the 18th-century chemical revolution and had a large influence on both the history of chemistry and the history of biology. He is widely considered in popular literature as the "father of modern chemistry".
It is generally accepted that Lavoisier's great accomplishments in chemistry largely stem from his changing the science from a qualitative to a quantitative one. Lavoisier is most noted for his discovery of the role oxygen plays in combustion. He recognized and named oxygen (1778) and hydrogen (1783) and opposed the phlogiston theory. Lavoisier helped construct the metric system, wrote the first extensive list of elements, and helped to reform chemical nomenclature. He predicted the existence of silicon (1787) and was also the first to establish that sulfur was an element (1777) rather than a compound. He discovered that, although matter may change its form or shape, its mass always remains the same.
Lavoisier was a powerful member of a number of aristocratic councils, and an administrator of the Ferme générale. The Ferme générale was one of the most hated components of the Ancien Régime because of the profits it took at the expense of the state, the secrecy of the terms of its contracts, and the violence of its armed agents. All of these political and economic activities enabled him to fund his scientific research. At the height of the French Revolution, he was accused by Jean-Paul Marat of selling adulterated tobacco and of other crimes, and was eventually guillotined a year after Marat's death.
Lavoisier's education was filled with the ideals of the French Enlightenment of the time, and he was fascinated by Pierre Macquer's dictionary of chemistry. He attended lectures in the natural sciences. Lavoisier's devotion and passion for chemistry were largely influenced by Étienne Condillac, a prominent French scholar of the 18th century. His first chemical publication appeared in 1764. From 1763 to 1767, he studied geology under Jean-Étienne Guettard. In collaboration with Guettard, Lavoisier worked on a geological survey of Alsace-Lorraine in June 1767. In 1764 he read his first paper to the French Academy of Sciences, France's most elite scientific society, on the chemical and physical properties of gypsum (hydrated calcium sulfate), and in 1766 he was awarded a gold medal by the King for an essay on the problems of urban street lighting. In 1768 Lavoisier received a provisional appointment to the Academy of Sciences. In 1769, he worked on the first geological map of France.
During late 1772 Lavoisier turned his attention to the phenomenon of combustion, the topic on which he was to make his most significant contribution to science. He reported the results of his first experiments on combustion in a note to the Academy on 20 October, in which he reported that when phosphorus burned, it combined with a large quantity of air to produce acid spirit of phosphorus, and that the phosphorus increased in weight on burning. In a second sealed note deposited with the Academy a few weeks later (1 November) Lavoisier extended his observations and conclusions to the burning of sulfur and went on to add that "what is observed in the combustion of sulfur and phosphorus may well take place in the case of all substances that gain in weight by combustion and calcination: and I am persuaded that the increase in weight of metallic calces is due to the same cause."
TEXT 6
Henry Cavendish
10 146 п.з.
Henry Cavendish (10 October 1731 – 24 February 1810) was a British natural philosopher, scientist, and an important experimental and theoretical chemist and physicist. Cavendish is noted for his discovery of hydrogen or what he called "inflammable air". He described the density of inflammable air, which formed water on combustion, in a 1766 paper "On Factitious Airs". Antoine Lavoisier later reproduced Cavendish's experiment and gave the element its name.
A notoriously shy man (it has been postulated that he was on the autism spectrum), Cavendish was nonetheless distinguished for great accuracy and precision in his researches into the composition of atmospheric air, the properties of different gases, the synthesis of water, the law governing electrical attraction and repulsion, a mechanical theory of heat, and calculations of the density (and hence the mass) of the Earth. His experiment to measure the density of the Earth has come to be known as the Cavendish experiment.
Henry Cavendish was born on 10 October 1731 in Nice, where his family was living at the time. His mother was Lady Anne Grey, fourth daughter of Henry Grey, 1st Duke of Kent, and his father was Lord Charles Cavendish, third son of William Cavendish, 2nd Duke of Devonshire. The family traces its lineage across eight centuries to Norman times and was closely connected to many aristocratic families of Great Britain. His mother died in 1733, three months after the birth of her second son, Frederick, and shortly before Henry’s second birthday, leaving Lord Charles Cavendish to bring up his two sons.
At age 11, Henry attended Hackney Academy, a private school near London. At age 18 (on 24 November 1748) he entered the University of Cambridge in St Peter's College, now known as Peterhouse, but left three years later on 23 February 1751 without taking a degree (a common practice). He then lived with his father in London, where he soon had his own laboratory.
Lord Charles Cavendish spent his life, first, in politics and then increasingly in science, especially in the Royal Society of London. In 1758, he took Henry to meetings of the Royal Society and also to dinners of the Royal Society Club. In 1760, Henry Cavendish was elected to both these groups, and he was assiduous in his attendance thereafter. He was active in the Council of the Royal Society of London (to which he was elected in 1765).
About the time of his father's death, Cavendish began to work closely with Charles Blagden, an association that helped Blagden enter fully into London’s scientific society. In return, Blagden helped to keep the world at a distance from Cavendish. Cavendish published no books and few papers, but he achieved much. Several areas of research, including mechanics, optics, and magnetism, feature extensively in his manuscripts, but they scarcely feature in his published work. Cavendish is considered to be one of the so-called pneumatic chemists of the eighteenth and nineteenth centuries, along with, for example, Joseph Priestley, Joseph Black, and Daniel Rutherford. Cavendish found that a definite, peculiar, and highly inflammable gas, which he referred to as "Inflammable Air" was produced by the action of certain acid on certain metals. This gas was in fact hydrogen, which Cavendish correctly guessed was proportioned to two in one water.
Although others, such as Robert Boyle, had prepared hydrogen gas earlier, Cavendish is usually given the credit for recognizing its elemental nature. Also, by dissolving alkalis in acids, Cavendish made "fixed air" (carbon dioxide), which he collected, along with other gases, in bottles inverted over water or mercury. He then measured their solubility in water and their specific gravity and noted their combustibility. Cavendish was awarded the Royal Society’s Copley Medal for this paper. Gas chemistry was of increasing importance in the latter half of the 18th century and became crucial for Frenchman Antoine-Laurent Lavoisier’s reform of chemistry, generally known as the chemical revolution.
In 1783 Cavendish published a paper on eudiometry (the measurement of the goodness of gases for breathing). He described a new eudiometer of his own invention, with which he achieved the best results to date, using what in other hands had been the inexact method of measuring gases by weighing them. He next published a paper on the production of water by burning inflammable air (that is, hydrogen) in "dephlogisticated air" (now known to be oxygen), the latter a constituent of atmospheric air (phlogiston theory).
Cavendish concluded that dephlogisticated air was dephlogisticated water and that hydrogen was either pure phlogiston or phlogisticated water. He reported these findings to Joseph Priestley, an English clergyman and scientist, no later than March 1783, but did not publish them until the following year. The Scottish inventor James Watt published a paper on the composition of water in 1783; Cavendish had performed the experiments first but published second. Controversy about priority ensured.
In 1785 Cavendish carried out an investigation of the composition of common (i.e. atmospheric) air, obtaining impressively accurate results. He conducted experiments in which hydrogen and ordinary air were combined in known ratios, and then exploded with a spark of electricity. Furthermore, he also described an experiment in which he was able to remove, in modern terminology, both the oxygen and nitrogen gases from a sample of atmospheric air until only a small bubble of unreacted gas was left in the original sample. Using his observations, Cavendish observed that, when he had determined the amounts of phlogisticated air (nitrogen) and dephlogisticated air (oxygen), there remained a volume of gas amounting to 1/120 of the original volume of nitrogen.
By careful measurements he was led to conclude that "common air consists of one part of dephlogisticated air [oxygen], mixed with four of phlogisticated [nitrogen]".
In the 1890s (around 100 years later) two British physicists, William Ramsay and Lord Rayleigh, realized that their newly discovered inert gas, argon, was responsible for Cavendish's problematic residue; he had not made an error. What he had done was perform rigorous quantitative experiments, using standardized instruments and methods, aimed at reproducible results; taken the mean of the result of several experiments; and identified and allowed for sources of error. The balance that he used, made by a craftsman named Harrison, was the first of the precision balances of the 18th century, and as accurate as Lavoisier's (which has been estimated to measure one part in 400,000). Cavendish worked with his instrument makers, generally improving existing instruments rather than inventing wholly new ones.
Cavendish, as indicated above, used the language of the old phlogiston theory in chemistry. In 1787 he became one of the earliest outside France to convert to the new antiphlogistic theory of Lavoisier, though he remained skeptical about the nomenclature of the new theory. He also objected to Lavoisier's identification of heat as having a material or elementary basis. Working within the framework of Newtonian mechanism, Cavendish had tackled the problem of the nature of heat in the 1760s, explaining heat as the result of the motion of matter.
In 1783 he published a paper on the temperature at which mercury freezes and in that paper made use of the idea of latent heat, although he did not use the term because he believed that it implied acceptance of a material theory of heat. He made his objections explicit in his 1784 paper on air. He went on to develop a general theory of heat, and the manuscript of that theory has been persuasively dated to the late 1780s. His theory was at once mathematical and mechanical: it contained the principle of the conservation of heat (later understood as an instance of conservation of energy) and even contained the concept (although not the label) of the mechanical equivalent of heat.
Cavendish's electrical and chemical experiments, like those on heat, had begun while he lived with his father in a laboratory in their London house. Lord Charles Cavendish died in 1783, leaving almost all of his very substantial estate to Henry. Like his theory of heat, Cavendish's comprehensive theory of electricity was mathematical in form and was based on precise quantitative experiments. In 1771 he published an early version of his theory, based on an expansive electrical fluid that exerted pressure. He demonstrated that if the intensity of electric force was inversely proportional to distance, then the electric fluid in excess of that needed for electrical neutrality would lie on the outer surface of an electrified sphere; then he confirmed this experimentally. Cavendish continued to work on electricity after this initial paper, but he published no more on the subject.
Cavendish wrote papers on electrical topics for the Royal Society but the bulk of his electrical experiments did not become known until they were collected and published by James Clerk Maxwell a century later, in 1879, long after other scientists had been credited with the same results. Cavendish’s electrical papers from the Philosophical Transactions of the Royal Society of London have been reprinted, together with most of his electrical manuscripts, in The Scientific Papers of the Honourable Henry Cavendish, F.R.S. (1921). According to the 1911 edition of Encyclopædia Britannica, among Cavendish's discoveries were the concept of electric potential (which he called the "degree of electrification"), an early unit of capacitance (that of a sphere one inch in diameter), the formula for the capacitance of a plate capacitor, the concept of the dielectric constant of a material, the relationship between electric potential and current (now called Ohm's Law) (1781), laws for the division of current in parallel circuits (now attributed to Charles Wheatstone), and the inverse square law of variation of electric force with distance, now called Coulomb's Law.
TEXT 7
James Clerk Maxwell
П.з.
James Clerk Maxwell (13 June 1831 – 5 November 1879) was a Scottish scientist in the field of mathematical physics. His most notable achievement was to formulate the classical theory of electromagnetic radiation, bringing together for the first time electricity, magnetism, and light as manifestations of the same phenomenon. Maxwell's equations for electromagnetism have been called the "second great unification in physics" after the first one realised by Isaac Newton.
With the publication of "A Dynamical Theory of the Electromagnetic Field" in 1865, Maxwell demonstrated that electric and magnetic fields travel through space as waves moving at the speed of light. Maxwell proposed that light is an undulation in the same medium that is the cause of electric and magnetic phenomena. The unification of light and electrical phenomena led to the prediction of the existence of radio waves.
Maxwell helped develop the Maxwell–Boltzmann distribution, a statistical means of describing aspects of the kinetic theory of gases. He is also known for presenting the first durable colour photograph in 1861 and for his foundational work on analysing the rigidity of rod-and-joint frameworks (trusses) like those in many bridges.
His discoveries helped usher in the era of modern physics, laying the foundation for such fields as special relativity and quantum mechanics. Many physicists regard Maxwell as the 19th-century scientist having the greatest influence on 20th-century physics. His contributions to the science are considered by many to be of the same magnitude as those of Isaac Newton and Albert Einstein. In the millennium poll—a survey of the 100 most prominent physicists—Maxwell was voted the third greatest physicist of all time, behind only Newton and Einstein. On the centenary of Maxwell's birthday, Einstein described Maxwell's work as the "most profound and the most fruitful that physics has experienced since the time of Newton".
Maxwell had studied and commented on electricity and magnetism as early as 1855 when his paper "On Faraday's lines of force" was read to the Cambridge Philosophical Society. The paper presented a simplified model of Faraday's work and how electricity and magnetism are related. He reduced all of the current knowledge into a linked set of differential equations with 20 equations in 20 variables. This work was later published as "On Physical Lines of Force" in March 1861.
Around 1862, while lecturing at King's College, Maxwell calculated that the speed of propagation of an electromagnetic field is approximately that of the speed of light. He considered this to be more than just a coincidence, commenting, "We can scarcely avoid the conclusion that light consists in the transverse undulations of the same medium which is the cause of electric and magnetic phenomena."
Working on the problem further, Maxwell showed that the equations predict the existence of waves of oscillating electric and magnetic fields that travel through empty space at a speed that could be predicted from simple electrical experiments; using the data available at the time, Maxwell obtained a velocity of 310,740,000 metres per second (1.0195×109 ft/s). In his 1864 paper "A Dynamical Theory of the Electromagnetic Field", Maxwell wrote, "The agreement of the results seems to show that light and magnetism are affections of the same substance, and that light is an electromagnetic disturbance propagated through the field according to electromagnetic laws".
His famous twenty equations, in their modern form of four partial differential equations, first appeared in fully developed form in his textbook A Treatise on Electricity and Magnetism in 1873. Maxwell expressed electromagnetism in the algebra of quaternions and made the electromagnetic potential the centrepiece of his theory. In 1881 Oliver Heaviside replaced Maxwell's electromagnetic potential field by 'force fields' as the centrepiece of electromagnetic theory. Heaviside reduced the complexity of Maxwell's theory down to four differential equations, known now collectively as Maxwell's Laws or Maxwell's equations. According to Heaviside, the electromagnetic potential field was arbitrary and needed to be "murdered". The use of scalar and vector potentials is now standard in the solution of Maxwell's equations.
A few years later there was a debate between Heaviside and Peter Guthrie Tait about the relative merits of vector analysis and quaternions. The result was the realisation that there was no need for the greater physical insights provided by quaternions if the theory was purely local, and vector analysis became commonplace. Maxwell was proven correct, and his quantitative connection between light and electromagnetism is considered one of the great accomplishments of 19th century mathematical physics.
Maxwell also introduced the concept of the electromagnetic field in comparison to force lines that Faraday described. By understanding the propagation of electromagnetism as a field emitted by active particles, Maxwell could advance his work on light. At that time, Maxwell believed that the propagation of light required a medium for the waves, dubbed the luminiferous aether. Over time, the existence of such a medium, permeating all space and yet apparently undetectable by mechanical means, proved impossible to reconcile with experiments such as the Michelson–Morley experiment. Moreover, it seemed to require an absolute frame of reference in which the equations were valid, with the distasteful result that the equations changed form for a moving observer. These difficulties inspired Albert Einstein to formulate the theory of special relativity; in the process Einstein dispensed with the requirement of a stationary luminiferous aether.
Maxwell also investigated the kinetic theory of gases. Originating with Daniel Bernoulli, this theory was advanced by the successive labours of John Herapath, John James Waterston, James Joule, and particularly Rudolf Clausius, to such an extent as to put its general accuracy beyond a doubt; but it received enormous development from Maxwell, who in this field appeared as an experimenter (on the laws of gaseous friction) as well as a mathematician.
Between 1859 and 1866, he developed the theory of the distributions of velocities in particles of a gas, work later generalised by Ludwig Boltzmann. The formula, called the Maxwell–Boltzmann distribution, gives the fraction of gas molecules moving at a specified velocity at any given temperature. In the kinetic theory, temperatures and heat involve only molecular movement. This approach generalised the previously established laws of thermodynamics and explained existing observations and experiments in a better way than had been achieved previously. Maxwell's work on thermodynamics led him to devise the thought experiment that came to be known as Maxwell's demon, where the second law of thermodynamics is violated by an imaginary being capable of sorting particles by energy.
In 1871 he established Maxwell's thermodynamic relations, which are statements of equality among the second derivatives of the thermodynamic potentials with respect to different thermodynamic variables. In 1874, he constructed a plaster thermodynamic visualisation as a way of exploring phase transitions, based on the American scientist Josiah Willard Gibbs's graphical thermodynamics papers.
TEXT 7